Pharmaceutical Logistics Compliance: What Specialty Pharmacies Need from a Last-Mile Courier
Pharmaceutical logistics compliance is usually discussed through the lens of manufacturers, wholesalers, distributors, warehouses, and freight networks. That matters, but it leaves a critical layer underexamined: what happens after a specialty medication leaves the pharmacy and moves toward the patient.
For specialty pharmacies, the last mile is where compliance becomes visible. It is where privacy, custody, temperature, patient access, and proof collide in one delivery event.
The stakes are high. Mercer projects that specialty drugs will account for over half of drug spend in 2025–2026 and beyond, which means delivery failures can carry real financial, operational, and patient-care consequences.
A courier partner is not just moving a package. In many specialty pharmacy workflows, the courier becomes part of the pharmacy’s compliance environment. This article explains what pharmaceutical logistics compliance means at the last-mile level, which risks matter most in direct-to-patient delivery, what ACHC and URAC expectations imply, and how to evaluate whether your courier partner can support audit-ready delivery operations.
What Pharmaceutical Logistics Compliance Means in Last-Mile Delivery
Pharmaceutical logistics compliance generally refers to the controls used to protect medication safety, integrity, traceability, and documentation as products move through the supply chain. In upstream logistics, that may include manufacturing controls, warehousing, distributor requirements, product serialization, and regulated storage.
At the last-mile level, pharmaceutical logistics requirements become more practical and more specific. The question shifts from “Was the product traceable through the supply chain?” to “Can the pharmacy prove the medication reached the right person, under the right conditions, with the right documentation?”
For specialty pharmacies, last-mile pharmaceutical delivery regulations and compliance expectations can touch several operating areas:
- Protecting patient information during delivery
- Maintaining chain of custody for sensitive or controlled medications, where applicable
- Preserving cold-chain or temperature-sensitive handling requirements
- Supporting REMS-related workflow needs
- Verifying delivery to the right recipient or approved handoff point
- Capturing proof of delivery
- Documenting exceptions, failed attempts, delays, damaged packages, or temperature concerns
This is a narrower view than broad pharma supply chain compliance. FDA’s Drug Supply Chain Security Act, for example, is designed to identify and trace certain prescription drugs at the package level as they move through the U.S. supply chain. That upstream traceability context matters, but specialty pharmacy last-mile delivery has its own practical control layer.Once the medication moves into direct-to-patient delivery, compliance depends on how well the courier workflow supports privacy, custody, temperature management, patient handoff, and documentation. That is why specialty pharmacies need delivery partners built for healthcare logistics, with workflows that account for documentation, handoff controls, and patient-sensitive exceptions.
Why Outsourcing Delivery Does Not Outsource Compliance Responsibility
A specialty pharmacy can outsource vehicles, couriers, routing, scheduling, tracking technology, and delivery execution. It cannot fully outsource accountability for whether medication reaches the patient safely, privately, and with adequate documentation.
That distinction matters.
If a medication is delayed, misdelivered, left with the wrong recipient, exposed to temperature risk, or documented poorly, the pharmacy may still need to answer for the process. A courier failure can become a pharmacy operations issue, a patient experience issue, an accreditation concern, or a payer confidence issue.
This is why specialty pharmacy last-mile delivery compliance should be treated as part of the pharmacy’s quality system, not as a simple vendor handoff.
From the patient’s perspective, the courier is often the final face of the pharmacy. From a compliance perspective, the courier is part of the record.
That means specialty pharmacies should evaluate courier partners the same way they evaluate other compliance-sensitive service providers. The review should go beyond rates and coverage maps. It should examine training, documentation, handoff controls, exception handling, oversight, auditability, and workflow fit.
For pharmacies building or improving direct-to-patient logistics, the delivery partner needs to support the compliance standards the pharmacy is accountable for. A low-cost courier that creates blind spots at the doorstep can become expensive quickly.
Key Direct-to-Patient Pharmaceutical Logistics Compliance Risks
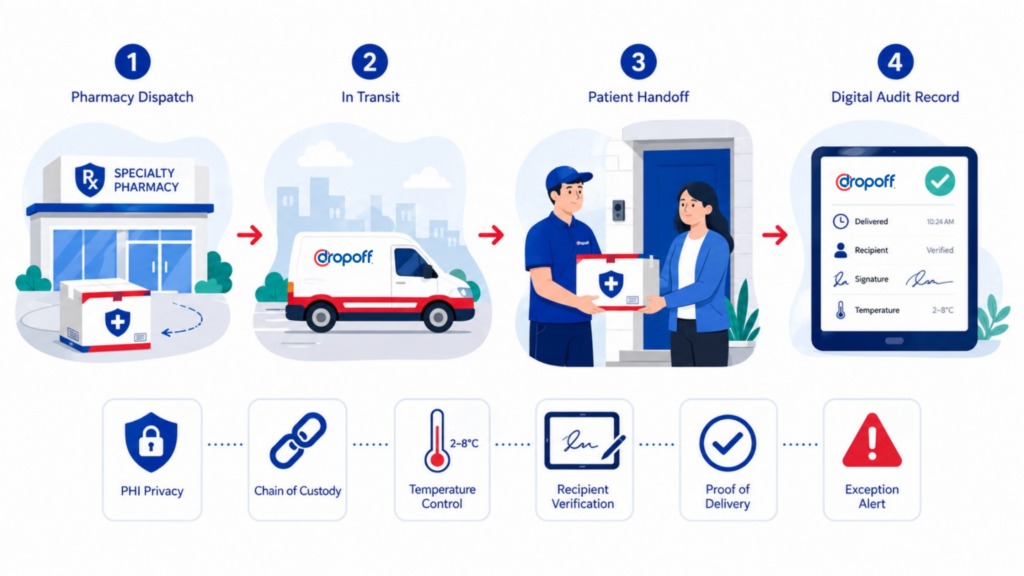
The most useful way to think about last-mile compliance is through delivery failure modes. What can go wrong between pharmacy dispatch and patient handoff? What records would prove what happened? What controls reduce the risk before it becomes an incident?
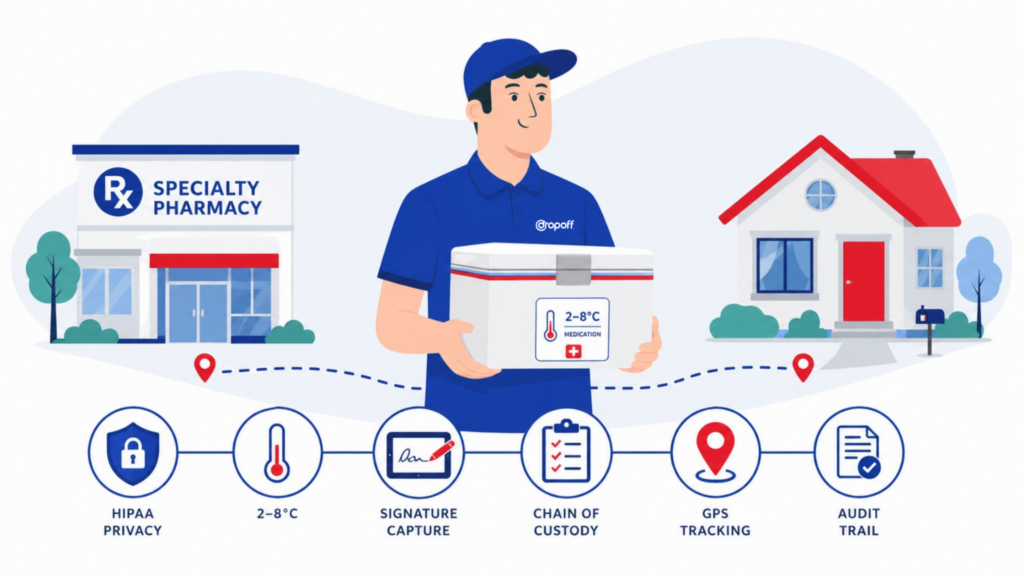
HIPAA and PHI Exposure During Delivery
Delivery workflows can expose patient information through package labels, notifications, delivery instructions, recipient conversations, support calls, or handoff notes. HHS explains that a business associate is an entity performing certain functions or services involving protected health information on behalf of a covered entity. See how HHS defines a business associate.
HHS also states that the Privacy Rule does not require business associate contracts with organizations such as the U.S. Postal Service or certain private couriers that act merely as conduits, when they transport information and access it only randomly or infrequently.
The practical takeaway: specialty pharmacies should evaluate whether a courier relationship involves PHI access or handling and whether a BAA is required. They should avoid assuming every courier relationship is automatically outside HIPAA risk. For more delivery-specific context, see Dropoff’s guide to HIPAA compliant medical courier practices.
Chain of Custody for Controlled or Sensitive Medications
Some specialty medications require tighter handoff controls, especially if they are high-value, clinically urgent, sensitive, or controlled. In those cases, the courier workflow should answer basic custody questions clearly.
Who picked up the medication? When was it picked up? Where did it travel? Who had possession during transit? Who received it? What happened if delivery failed?
For medications subject to controlled-substance handling requirements, specialty pharmacies should confirm that courier handoffs, recipient verification, and exception records align with internal policies and applicable DEA-related obligations.
Cold Chain and Temperature-Sensitive Medication Handling
Specialty pharmacies often handle medications with specific temperature requirements. Last-mile risk can happen during dwell time, traffic delays, failed first attempts, incorrect delivery locations, long handoff windows, or poorly managed exceptions.
URAC notes that pharmacy accreditation standards have expanded to address medication delivery and internal storage procedures at all temperature levels, and that organizations must qualify product storage and delivery programs through continuous monitoring and testing. ACHC also states that pharmacy accreditation standards require organizations to ship pharmaceuticals in a manner that maintains appropriate sanitation, light, and temperature conditions.
That makes pharmacy courier cold chain compliance a delivery operations issue, not just a packaging issue. For specialty pharmacies managing temperature-sensitive medications, courier workflows need to support route timing, handoff windows, exception response, and documentation across the full cold chain logistics in healthcare workflow.
REMS Program Delivery Requirements
REMS programs are FDA-required safety programs for certain medications with serious safety concerns. FDA says REMS are designed to reinforce medication use behaviors and actions that support safe use.
Some REMS requirements involve specific participants, settings, or dispensing controls. FDA also notes that some REMS require that a pharmacy or healthcare setting be certified to receive or dispense the medication.
Couriers do not manage REMS programs for pharmacies. But for REMS-related therapies, the delivery workflow should support the pharmacy’s obligations through reliable timing, recipient controls, documentation, and exception visibility. A missed delivery, unclear handoff, or poor record can create unnecessary friction in an already controlled process.
Proof of Delivery and Recipient Verification
“Delivered” is not always enough.
Specialty pharmacies may need documentation showing when delivery occurred, where it occurred, who accepted it, and whether a signature or identity confirmation was captured. Proof of delivery should reduce ambiguity. It should not simply mark a package complete.
Useful proof of delivery may include a timestamp, GPS or location data, signature capture, recipient name, photo confirmation where appropriate, delivery notes, and exception history. The exact requirements will vary by medication, pharmacy policy, and patient workflow, but the principle is consistent: the record should be strong enough to review later.
Failed Attempts, Exceptions, and Audit Trails
Exceptions are where weak delivery programs become visible.
A failed attempt, patient unavailable event, address mismatch, route delay, temperature concern, damaged package, or partial documentation issue should create a clear record. Specialty pharmacies need to know what happened, when it happened, who was involved, how it was escalated, and how the issue was resolved.
Screenshots, scattered phone calls, and incomplete support notes are fragile records. Audit-ready courier workflows should make exceptions visible, traceable, and retrievable.
What ACHC and URAC Expectations Mean for Last-Mile Pharmaceutical Delivery
Accreditation expectations turn delivery from a vendor preference into a quality management issue. Specialty pharmacies need to show that medication storage, shipping, and delivery processes protect product integrity, patient safety, and documentation quality.
URAC’s temperature-management guidance is especially relevant for last-mile delivery because it connects accreditation expectations to storage and delivery qualification. URAC says organizations must qualify product storage and delivery programs through continuous monitoring and testing. That has practical implications for how specialty pharmacies validate packaging, route timing, temperature control, delivery monitoring, and response procedures.
ACHC’s pharmacy guidance also emphasizes shipping pharmaceuticals in a way that maintains appropriate sanitation, light, and temperature conditions. That means the delivery process has to support the medication’s handling requirements from dispatch through handoff.
For specialty pharmacies, these expectations translate into concrete courier requirements:
- Documented shipping and delivery processes
- Temperature-capable workflows
- Validated packaging or handling procedures, where applicable
- Delivery monitoring
- Incident and exception logs
- Oversight of contracted service providers
- Retrievable delivery records
- Clear escalation paths for delays, failed attempts, and temperature concerns
Accreditation bodies are not simply asking whether medication was shipped. They are asking whether the organization can show that the shipping and delivery process protects product integrity, patient safety, and documentation quality.
That makes the courier part of accreditation readiness. A pharmacy courier should support the records, controls, and visibility the pharmacy may need during internal reviews, accreditation surveys, payer conversations, or quality investigations.If you are comparing providers, start with the basic medical courier service requirements that matter most in healthcare delivery: training, reliability, documentation, tracking, and support.
What to Look for in a Pharmaceutical Logistics Provider for Last-Mile Compliance

The right pharmaceutical logistics provider should do more than move medication quickly. For specialty pharmacy delivery, the provider should help support privacy, traceability, temperature-sensitive handling, recipient verification, and audit-ready documentation.
Use the criteria below when evaluating pharmaceutical logistics providers compliance for last-mile delivery.
HIPAA BAA Readiness When Required
A courier partner should be able to support appropriate contracting if the relationship involves PHI handling or service functions requiring a business associate agreement. The courier should understand healthcare delivery privacy expectations and avoid casual handling of patient-related information.
Background-Checked and Trained Drivers
Specialty pharmacy deliveries should not be treated like generic gig delivery coverage. Drivers should be vetted, background-checked, and trained for healthcare-sensitive delivery expectations, including professionalism, patient privacy, handoff procedures, and exception escalation.
Temperature-Controlled or Temperature-Sensitive Delivery Capabilities
A courier should be able to support medication-specific delivery windows, handling requirements, and escalation procedures. For cold-chain or temperature-sensitive medications, pharmacy courier cold chain compliance depends on how the workflow manages timing, dwell time, failed attempts, and documentation.
Real-Time Tracking That Supports Traceability
Tracking should show more than a consumer-style map pin. Specialty pharmacies need visibility into pickup, transit, delivery status, route progress, timing, and exceptions. Strong last-mile delivery tracking helps pharmacy teams see whether a delivery is on track, delayed, or at risk.
Signature Capture and Recipient Verification
For sensitive deliveries, the pharmacy should know whether the medication reached the intended recipient or approved handoff point. Signature capture, recipient name, location confirmation, and delivery notes can help reduce uncertainty.
Audit-Ready Proof of Delivery
Delivery records should include the details a pharmacy may need later: timestamp, location, driver notes, signature or photo where appropriate, recipient information, and exception history. Strong proof of delivery for business deliveries creates a cleaner record than a simple completion status.
Exception Management and 24/7 Support
Urgent medication deliveries need a process when something goes wrong. Patient unavailable. Route delay. Damaged package. Failed handoff. Temperature concern. Address mismatch.
The courier should have clear escalation procedures and responsive support, especially for time-sensitive healthcare deliveries.
Specialty Pharmacy Fit
The best courier is not merely fast. It can adapt to pharmacy workflows, direct-to-patient requirements, patient-sensitive communication, compliance documentation, route design, and delivery program needs.
For specialty pharmacies, specialty pharmacy delivery solutions should feel like an extension of the pharmacy’s operating model, not a generic transportation add-on.
How to Evaluate Your Current Courier Against Pharmaceutical Logistics Compliance Requirements
Before your next courier renewal, accreditation review, or direct-to-patient program expansion, evaluate whether your current delivery partner can support your pharmaceutical logistics compliance requirements.
Use this mini audit as a starting point:
| Compliance Area | What to Verify | Why It Matters |
| Pickup and custody | Can the courier document pickup, driver assignment, transit status, handoff, and completion? | Creates a clearer chain of custody and delivery record. |
| Audit trail | Can records be retrieved quickly during an audit, payer review, or accreditation process? | Reduces scramble when documentation is requested. |
| HIPAA and PHI | Can the courier support HIPAA-related contracting where required? | Helps address privacy risk when delivery workflows involve PHI. |
| Driver standards | Are drivers background-checked and trained for healthcare delivery expectations? | Protects professionalism, patient trust, and sensitive handoffs. |
| Temperature-sensitive delivery | Can the courier support medications with specific timing or handling requirements? | Helps reduce cold-chain and temperature excursion risk. |
| Proof of delivery | Does delivery confirmation include more than “delivered”? | Supports recipient verification and issue resolution. |
| Exception handling | How are failed attempts, delays, damage, or temperature concerns escalated? | Keeps problems from disappearing into support fog. |
| Support coverage | Does the courier offer support outside standard business hours? | Matters for urgent, scheduled, or patient-critical deliveries. |
| Workflow fit | Does the courier understand specialty pharmacy workflows? | Helps align delivery execution with patient care and compliance needs. |
This evaluation can also support a broader vendor review. The goal is to assess whether your courier can meet the operational demands of specialty pharmacy delivery, including documentation, visibility, escalation, patient-sensitive handling, and compliance support.
How Dropoff Supports Specialty Pharmacy Last-Mile Delivery Compliance
Dropoff supports healthcare and pharmacy delivery workflows with professional couriers, real-time tracking, digital confirmations, customizable delivery programs, and responsive support. For specialty pharmacies, that means more visibility and documentation around direct-to-patient delivery.
Dropoff’s model is designed to help support specialty pharmacy last-mile delivery compliance through:
- Vetted professional drivers
- Real-time tracking
- Digital delivery confirmations
- Chain-of-custody support
- Scheduled, on-demand, route, and express delivery options
- Healthcare delivery experience
- 24/7 support
This matters because specialty pharmacy delivery is rarely just a transportation problem. It often involves patient availability, sensitive medications, documentation needs, timing requirements, and exception management.
Before your next accreditation review or courier renewal, evaluate whether your delivery partner can produce the records, visibility, and exception controls your pharmacy depends on. Dropoff’s pharmacy courier services are built to help pharmacies strengthen direct-to-patient delivery programs with more control, visibility, and reliability.
For broader healthcare delivery needs, explore Dropoff’s same-day healthcare delivery solutions.
FAQs About Pharmaceutical Logistics Compliance in Specialty Pharmacy Delivery
Pharmaceutical logistics compliance refers to the controls used to protect medication integrity, safety, traceability, privacy, and documentation as pharmaceuticals move from one point to another. In direct-to-patient specialty pharmacy delivery, it includes custody, temperature control, recipient verification, HIPAA and PHI considerations, proof of delivery, and exception documentation.
Yes. In practical compliance terms, the pharmacy still needs confidence that medications are delivered safely, privately, and with adequate documentation. A courier may handle the physical delivery, but the pharmacy often remains accountable for the quality, reliability, and records associated with the delivery process. The courier becomes part of the pharmacy’s delivery controls.
It depends on the courier’s role and PHI exposure. HHS recognizes a conduit concept for certain couriers that only transport information and access it randomly or infrequently. However, specialty pharmacies should evaluate whether a courier relationship involves PHI access, handling, systems, or services that require a BAA or other HIPAA-related controls.
Pharmacy courier cold chain compliance depends on more than packaging. Temperature-sensitive delivery requires appropriate handling, delivery timing, minimized dwell time, clear escalation procedures, and documentation. Specialty pharmacies should verify whether their courier can support the medication’s requirements and provide records if a delay, failed delivery, or temperature concern occurs.
Specialty pharmacies should look for HIPAA BAA readiness where applicable, vetted drivers, healthcare delivery experience, real-time tracking, temperature-sensitive delivery capabilities, signature capture, audit-ready proof of delivery, exception management, and responsive support. The courier should understand specialty pharmacy workflows, not just parcel delivery.
Make Your Last-Mile Courier Part of Your Compliance Program
Pharmaceutical logistics compliance is not only about upstream supply chain controls. For specialty pharmacies, the last mile is where privacy, custody, temperature, patient access, and documentation come together.
A specialty pharmacy courier should strengthen your compliance posture, not create another blind spot. The right partner should help you see what happened, prove what happened, and respond quickly when something changes.If your current courier cannot support the records, visibility, and handoff controls your pharmacy depends on, it may be time to reassess the delivery program. Explore Dropoff’s specialty pharmacy courier services to build a more reliable, documented, and patient-ready last-mile delivery operation.