10 Best Clinical Trial Logistics Companies to Use in 2026

A shipment can arrive on time and still fail the trial.
That is the real problem with many lists of clinical trial logistics companies. They treat logistics like transport, when clinical teams know it is closer to protocol execution. The question is not only whether a package arrived. It is whether it arrived in the right condition, inside the right timing window, with the right documentation, and without creating extra work for the site, lab, patient, or sponsor.
The stakes are getting higher. Grand View Research estimates the U.S. clinical trials supply and logistics market was $1.40 billion in 2024 and could reach $2.87 billion by 2033. ClinicalTrials.gov also shows registered studies rising from 477,192 in 2024 to 520,863 in 2025. As trials become more complex, sponsor teams need clinical logistics companies that can do more than move boxes.
The best clinical trial logistics companies are not all built for the same job. Some are strongest in global trial supply, some in specialty cold chain, some in decentralized support, and some in high-control U.S. last-mile execution. This guide explains how to evaluate providers, then compares the top options by use case.
Quick decision tool: which type of clinical trial logistics provider fits your study?
Use this before comparing vendors. The right choice usually depends less on who has the biggest network and more on where your trial is most likely to break.
| If your biggest need is… | You likely need… | Best-fit provider type |
| Global trial supply coordination across countries, depots, and sites | End-to-end orchestration, customs support, broad infrastructure, and cross-border control | Global clinical trial logistics provider |
| Cryogenic shipping or advanced therapy handling | Deep temperature-control expertise, chain-of-condition support, and specialty cold-chain processes | Specialty cold-chain provider |
| Direct-to-patient or decentralized trial support | Patient-facing delivery workflows, failed-attempt handling, and home-based shipment coordination | Decentralized / patient-centric provider |
| U.S. site-to-lab specimen movement with narrow timing windows | Same-day responsiveness, strong handoff control, proof of custody, and exception visibility | U.S. last-mile clinical logistics partner |
| Packaging, labeling, and broader clinical supply support | Clinical packaging, forecasting, depot distribution, and compliance-heavy supply services | Clinical supply services provide |
| Europe-heavy regional trial execution | Temperature-controlled healthcare distribution with stronger regional focus | Europe-focused specialist provider |
Rule of thumb: match the provider to your main failure risk. If your study is most vulnerable to customs delays, depot coordination, and multi-country complexity, start with a global end-to-end operator. If your risk sits in specimen viability, timing windows, handoff integrity, and U.S. execution, a specialized last-mile partner may be the better fit.
What do clinical trial logistics companies actually handle?
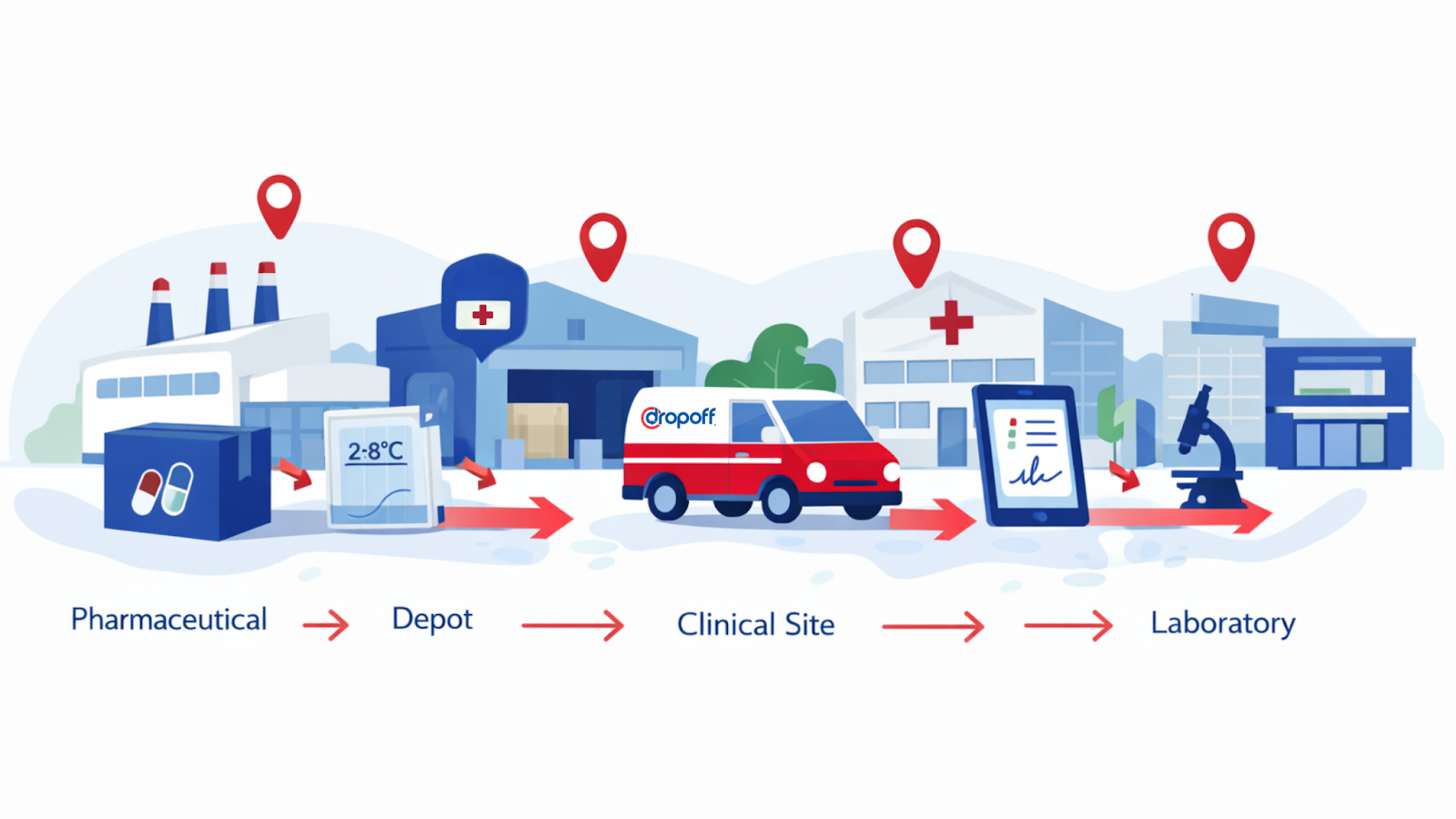
At a practical level, clinical trial logistics covers much more than shipping. Depending on the study, providers may be responsible for direct-to-site investigational product distribution, site-to-lab specimen movement, comparator sourcing support, depot coordination, temperature-controlled shipping, returns, resupply, packaging, labeling support, and exception handling across multiple stakeholders.
The best clinical trial logistics services are built around protocol-specific handling requirements rather than generic healthcare freight. That matters even more in decentralized trials. FDA’s guidance on decentralized elements explicitly addresses shipping, receipt, tracking, participant safety, and documentation when trial activities happen away from traditional sites. MarketsandMarkets also says North America accounted for 41.4% of clinical trial supplies market revenue in 2024, helped by decentralized trial adoption and advanced cold-chain logistics.
In real life, one provider may be handling investigational products on one leg, biological samples on another, and patient-facing shipments somewhere else entirely. Some companies are built for global clinical supply orchestration. Some are strongest in cryogenic or specialty temperature-controlled workflows. Some lean into direct-to-patient support. Others are better at regional execution, where same-day movement, handoff integrity, and visibility matter more than sheer network breadth.
That is why provider fit matters so much. A company that excels at global depot strategy may not be the best fit for urgent site-to-lab movement. A company with mature direct-to-patient processes may be more useful for hybrid or decentralized studies than for a conventional multi-country depot model. The right choice depends on trial design, geography, sample sensitivity, and how much control your team needs at the last mile.
How to evaluate clinical trial logistics companies before you sign
The criteria should come first, because the right provider for a cell and gene therapy study, a decentralized patient-facing trial, and a U.S. regional site-to-lab workflow may be three different kinds of company. That matters more now because trial complexity is rising. A 2024 Scientific Reports analysis of more than 16,000 trials found that clinical trials have become more complex over time. The same study linked that complexity to greater burden and longer timelines.
2. Chain of custody and documentation
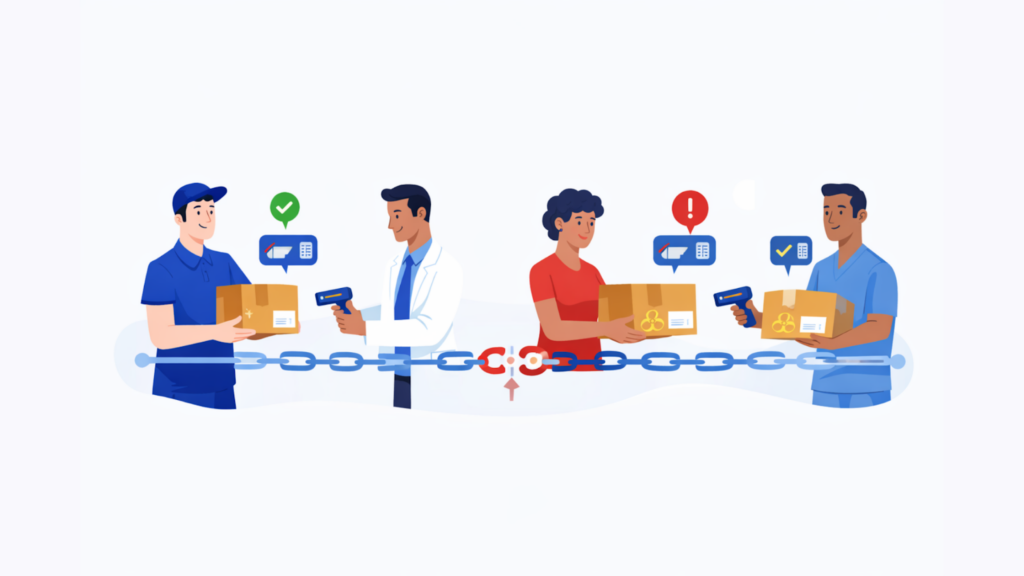
Start with traceability.
You want to know how the provider records scan events, captures proof of delivery, logs exceptions, and preserves an audit trail. Visibility is useful. Traceability is better. A tracking screen that says “delivered” does not tell you who accepted the shipment, whether custody changed hands correctly, or whether there was a gap between arrival and controlled intake.
In clinical workflows, that missing detail can matter more than the linehaul itself.
2. Temperature control and specimen integrity
“Cold chain capable” is too vague to be reassuring.
Ask what temperature ranges a provider supports routinely, how packaging is validated, what monitoring is used in transit, how excursions are handled, and how time out of range is documented. Controlled ambient, refrigerated, frozen, and cryogenic are different operating models, not different labels for the same thing.That is especially true for biological sample courier services, where timing, condition, and custody gaps can all affect whether a specimen remains usable. Anyone responsible for transporting blood and specimens already knows the risk is rarely one dramatic collapse. It is usually a stack of smaller misses across packaging, handoff, and intake timing.
3. Timing windows and protocol sensitivity
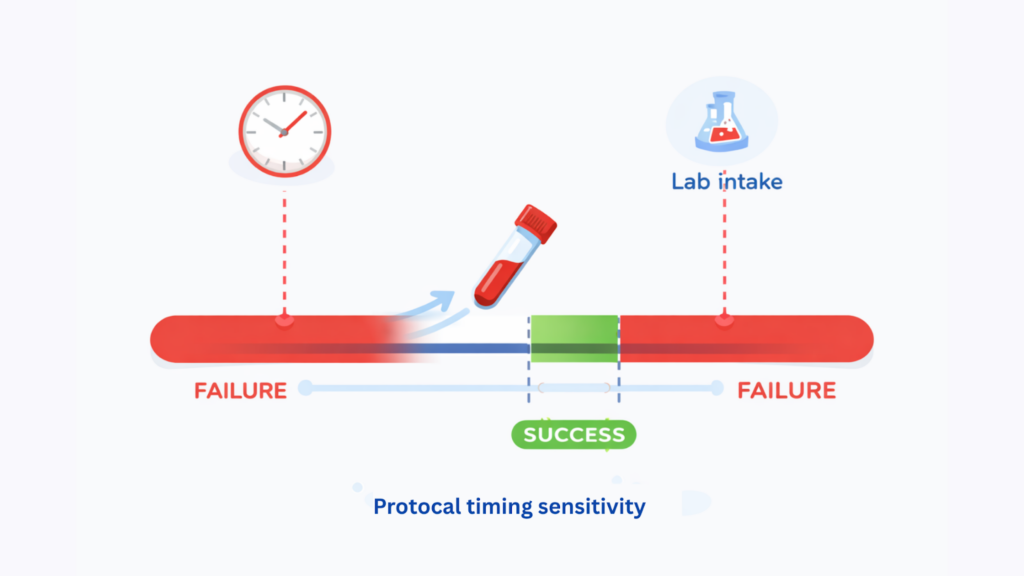
Clinical trial logistics is often a timing problem disguised as a transport problem.
Site cutoff times, lab intake windows, patient collection schedules, and same-day or STAT requirements all change the risk profile. A shipment can move quickly overall and still miss the only window that mattered. That is why “delivered” is not the same as “usable.”
4. Direct-to-site vs. direct-to-patient vs. site-to-lab fit
Not every provider is built for every model.
Direct-to-patient support involves participant communication, failed-attempt handling, and local coordination that look very different from depot-to-site replenishment. Site-to-lab specimen movement has its own discipline around collection timing, packaging, and handoff quality. If your study includes decentralized elements, make sure the provider’s operating model actually matches them. FDA’s decentralized-trial guidance makes that distinction more important, since shipping and distribution requirements shift once trial activity moves beyond the conventional site model.
5. Exception management and communication
Ask what happens when things go wrong.What if a site is closed? What if customs stalls a shipment? What if a participant is unavailable? What if a shipment arrives but no trained person accepts it? Clean-case performance is table stakes. The best clinical trial courier services earn their keep in exception response.
6. Coverage model and operating control
Finally, look at how the network really works.
A broad global footprint is valuable, but so is understanding when a company self-performs, when it hands off to partners, and where a specialized regional layer may add control. Global breadth and local execution are not the same strength.
A useful rule of thumb: choose the provider type that best matches your failure risk. If your trial risk sits in customs, depot coordination, and multi-country complexity, you may need a global end-to-end operator. If your risk sits in specimen timing, last-mile handoffs, and visibility into usable condition, a more execution-focused partner can matter just as much.
Top clinical trial logistics companies at a glance
Before the longer breakdown, here is the quick view:
| Company | Category | Best for | Geographic strength | Core capabilities | Decentralized / DTP support |
| Dropoff | U.S. last-mile execution | U.S. last-mile execution and specimen-sensitive workflows | U.S. regional / national | Same-day and scheduled healthcare logistics, proof of handoff, visibility, routed execution | Selective / workflow-dependent |
| DHL Life Sciences & Healthcare | U.S. and global execution | Broad clinical trial infrastructure with U.S. reach | Global | IMP storage and distribution, sample logistics, temperature monitoring, DTP support | Present |
| Marken | Global end-to-end | Global clinical supply plus patient-centric support | Global | Clinical supply chain, depots, labs, DTP/DFP | Strong |
| World Courier | Global end-to-end | Global specialty clinical trial logistics | Global | GxP depots, comparator sourcing, specialty transport, visibility | Strong |
| Almac | Specialty supply chain | Clinical supply strategy plus specimen logistics | Global | Distribution, depot services, specimen collection logistics, kit support | Moderate |
| Cryoport Systems | Specialty cold chain | Cryogenic and advanced therapy logistics | Global specialist | Cryogenic transport, biostorage, chain-of-condition support | Targeted |
| Catalent Clinical Supply Services | Specialty supply chain | Clinical supply planning and global distribution | Global | Packaging, forecasting, planning, depot distribution | Moderate |
| Parexel Clinical Logistics | Decentralized / patient-centric | Logistics integrated with CRO operations | Global | Regulatory, lab/sample management, drug supply, ancillaries | Present |
| Movianto | Decentralized / regional Europe | Europe-forward clinical trial logistics | Europe-focused | IMP storage, temperature-controlled distribution, returns, DTP support | Present |
| PCI Pharma Services | Packaging / compliance-heavy | Clinical support within a broader pharma-services model | Global | Clinical trial services, packaging, compliance depth | Moderate |
Best fit always depends on trial design, sample sensitivity, geography, and the level of control you need over handoffs and exceptions.
10 best clinical trial logistics companies to use in 2026
Clinical trial logistics companies by category
A flat top-10 list makes this category look simpler than it is. It isn’t. The best provider depends on whether your biggest risk sits in U.S. site-to-lab execution, specialty temperature control, decentralized trial support, or global depot coordination. That is why this roundup is organized by use case instead of pretending every company is solving the same operational problem.
Best for U.S. last-mile execution and specimen-sensitive workflows
Dropoff
Dropoff is the strongest fit when the real risk sits in U.S. execution, not global depot strategy. For healthcare-adjacent and specimen-sensitive workflows, the value is tighter control over handoffs, same-day and scheduled movement, proof of handoff, visibility, and regional execution discipline. Dropoff is especially relevant when teams need high-control U.S. site-to-lab movement, time-sensitive pickups, and workflows where “arrived” means very little unless the chain of custody holds all the way through.
DHL Life Sciences & Healthcare
DHL is broader than a pure U.S. last-mile specialist, but it still belongs in this category because of its scale, U.S. operational presence, and ability to support clinical trial infrastructure across multiple execution models. Its offering is more useful when sponsors need broad reach, established infrastructure, and integration across domestic and international movements rather than a narrower handoff-control play.
Best global end-to-end clinical trial logistics providers
Marken
Marken is strongest when a study needs a broad global clinical supply chain plus patient-centric support. It is well suited for multinational trials where site, lab, and patient workflows need to connect through one specialist network.
World Courier
World Courier stands out for specialty global clinical trial logistics. It is especially relevant for cross-border studies where storage discipline, specialty handling, and international coordination are central to the operating model.
Best for specialty cold chain and complex supply chains
Cryoport Systems
Cryoport is the specialist choice when the workflow is unusually temperature-sensitive, especially in cell and gene therapy or other advanced-therapy settings where chain of condition and cryogenic control matter heavily.
Almac
Almac is a strong option when clinical supply planning, specimen logistics, and kit management need to work together rather than sit in separate operational silos.
Catalent Clinical Supply Services
Catalent fits best when sponsors want packaging, planning, and global distribution tied together inside a broader clinical supply-services model.
Best for decentralized or patient-centric trial support
Parexel Clinical Logistics
Parexel is strongest when logistics needs to sit close to broader clinical execution. Its positioning makes the most sense for sponsors who want logistics, regulatory, and trial operations working in tighter alignment.
Movianto
Movianto is a strong contender for Europe-heavy programs that need healthcare-specialist distribution, temperature control, and support for patient-facing or regional trial workflows.
PCI Pharma Services
PCI belongs here as a broader clinical and pharma-services player with strong packaging and compliance depth, though buyers should verify exact logistics fit carefully depending on the study model.
When you need a specialized last-mile clinical logistics partner
A global operator can be the right answer for study-wide infrastructure. That does not mean it is the complete answer for every workflow inside the study.
Last-mile risk shows up in smaller, quieter places: specimen pickup timing, site-to-lab transfer discipline, handoff integrity, participant availability, intake mismatches, and the difference between a status update and a usable sample. As decentralized elements expand and protocol complexity increases, execution detail carries more weight. FDA’s guidance reinforces this by treating shipping, receipt, oversight, and documentation as critical parts of decentralized trial execution rather than administrative leftovers.
That is when a specialized last-mile layer starts to matter. If your workflow depends on narrow timing windows, specimen viability, local responsiveness, or precise exception response, you may need more control than a broad network abstraction can give you. The logic is similar to choosing a medical courier more broadly: the right fit depends less on broad claims and more on whether the operator can hold up under real-world handoffs, timing pressure, and documentation demands.
A specialized partner will not replace end-to-end clinical supply management. It will strengthen the part most likely to fail quietly.
7 questions to ask before choosing a clinical trial logistics company
- How do you maintain chain of custody across every handoff?
Ask for specifics: scan events, named acceptance, escalation records, and audit-trail depth. - What temperature ranges do you routinely support, and how do you handle excursions?
Routine capability matters more than vague claims. - How do you manage failed delivery attempts, delayed site acceptance, or participant unavailability?
Exception response is part of the service, not a side issue. - Do you support direct-to-patient, direct-to-site, site-to-lab, or all three?
Many providers are stronger in one model than another. - What real-time visibility will our team actually have?
Can you see custody, condition, and exception status, or just milestone scans? - When do you self-perform versus rely on partners?
This helps you understand where control may thin out. - Can you support protocol-specific workflows and narrow timing windows?
That includes site cutoffs, patient scheduling, special packaging, and documentation discipline.
Red flags include vague answers on excursions, visibility that stops at “delivered,” no clear escalation path, broad national or global claims without operating detail, and no clear distinction between standard medical courier work and trial-specific workflows. Many of the same principles behind the best questions to ask when choosing a medical courier apply here too, just with tighter protocol and documentation stakes.
Choosing the right clinical trial logistics company comes down to fit
The best clinical trial logistics company is not the one with the biggest logo wall. It is the one that matches the shape of your trial.
If your study depends on global orchestration, customs navigation, depot strategy, and broad specialty infrastructure, a large end-to-end provider may be the right choice. If your risk sits in cryogenic control, patient-centric distribution, or Europe-heavy execution, the shortlist shifts. And if your real operational exposure lives in U.S. handoffs, specimen visibility, and protocol-sensitive last-mile timing, specialized execution deserves a bigger role in the decision.
Teams that need tighter control over specimen-sensitive handoffs and U.S. execution can learn more about Dropoff’s clinical trial logistics services.
FAQs about clinical trial logistics companies
A clinical trial logistics company manages the movement, control, and documentation of trial-related materials such as investigational products, samples, kits, and returns across sites, labs, depots, patients, and other stakeholders.
Clinical logistics companies may coordinate direct-to-site distribution, site-to-lab specimen transport, temperature-controlled shipping, packaging and labeling support, chain-of-custody documentation, returns, resupply, and decentralized or direct-to-patient movement where the trial model requires it. FDA guidance makes clear that shipping, tracking, participant safety, and documentation all become more important as decentralized elements grow.
Usually through validated packaging, temperature-appropriate transport, documented handoffs, and time-sensitive movement tied to protocol requirements and lab intake windows. The risk is not just delay. It is delay plus condition plus documentation. In many studies, that means working with biological sample courier services that can support validated packaging, documented handoffs, and timing-sensitive specimen movement.
Some do, some do not, and support levels vary. FDA’s 2024 guidance makes clear that shipping, tracking, receipt, and participant safety remain central when investigational products or materials move beyond traditional sites. That means logistics capability for decentralized trials needs more than delivery capacity alone.
Look for fit across chain of custody, temperature control, timing-window discipline, exception handling, visibility, and the actual operating model behind the network. The best provider is the one aligned to your trial’s real failure risk.
Not always. There is overlap, especially around healthcare handling and documentation, but clinical trial courier services often need tighter protocol alignment, more specialized packaging and temperature handling, clearer audit trails, and closer coordination with sites, labs, depots, and patient-facing workflows.