Clinical Trial Logistics Explained: Processes, Challenges, and Best Practices
Clinical trial logistics is the operational framework that ensures investigational products (IP/IMP), lab kits, biological specimens, and ancillary supplies move through a study safely, compliantly, and on schedule. More than transportation, clinical trial logistics management governs temperature control, chain-of-custody documentation, site coordination, and audit readiness across the entire trial lifecycle.
When clinical trial logistics is structured and measurable, protocols stay intact, patient dosing windows are protected, and clinical data remains valid. When it breaks down, missed delivery windows, temperature excursions, and incomplete documentation can trigger protocol deviations, product replacement, or regulatory findings.
The operational stakes are rising. Trials now span more sites, incorporate decentralized components, and rely on temperature-sensitive biologics. According to IQVIA’s Global Trends in R&D 2025 report, clinical development cycle times and transition efficiency between phases continue to evolve as complexity increases. Even as macro-level efficiency improves, execution risk at the site and last-mile level remains a critical determinant of whether activation timelines stay intact.
While regulatory and site factors contribute to those delays, logistics failures — missed pickups, packaging errors, excursion events, incomplete proof-of-delivery — compound activation risk and extend time to first patient in.
For Clinical & Healthcare Supply Chain leaders, this is a governance issue. For Logistics and Transportation executives, it is an execution issue. In both cases, the central question is the same:
Is your clinical trial logistics model resilient enough to protect compliance, patient safety, and study timelines under real-world operating conditions?
The sections that follow break down the processes, risks, and measurable controls that define high-performing clinical research logistics programs.
What Is Clinical Trial Logistics?
Clinical trial logistics is the coordinated management of investigational products (IP/IMP), laboratory kits, biological specimens, and clinical supplies throughout a study lifecycle. It ensures temperature control, chain-of-custody documentation, regulatory compliance, and on-time delivery between manufacturers, depots, clinical sites, laboratories, and patients.
Clinical trial logistics management extends beyond transportation. It governs investigational product distribution, specimen transport, temperature monitoring, audit-ready documentation, and exception control across multi-site and decentralized clinical research environments.
Clinical research logistics and clinical study logistics are often used interchangeably, but this discipline differs from standard medical courier services. Traditional healthcare delivery focuses on speed and HIPAA compliance. Clinical trial logistics adds protocol adherence, blinding protection, chain-of-identity safeguards, essential record retention, and sponsor oversight into every transfer.
Key Stakeholders in Clinical Trial Logistics
Clinical trial logistics typically involves coordination among:
- Sponsor
- Contract Research Organization (CRO)
- Manufacturing partner
- Depot or central distribution hub
- Clinical site
- Central laboratory
- Specialized logistics provider
Each transfer point introduces operational and compliance risk. Every handoff must be documented, verified, and auditable.
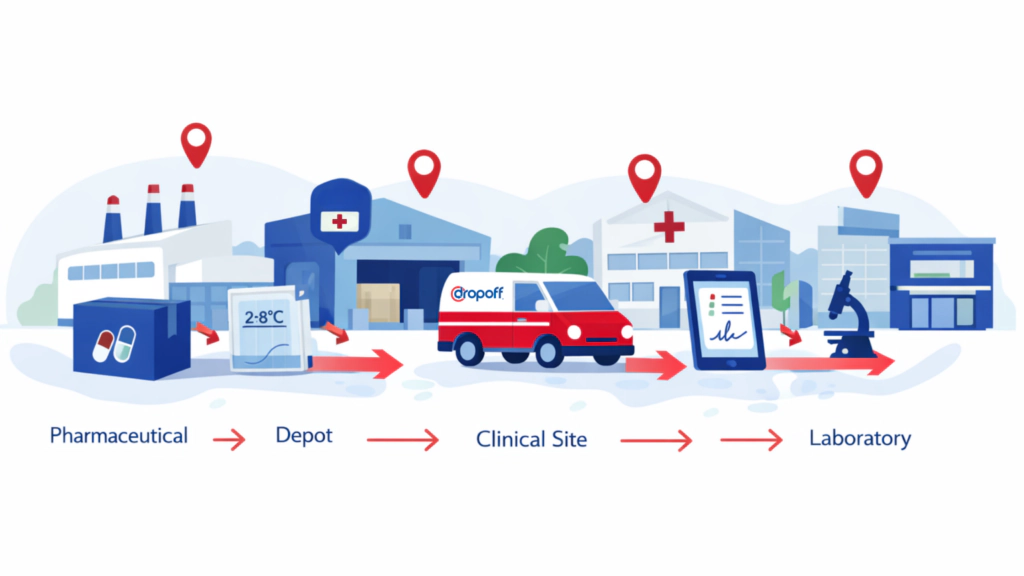
End-to-End Clinical Trial Logistics Flow
A simplified clinical trial logistics flow includes:
- Manufacturer → Depot
- Depot → Clinical Site
- Clinical Site → Central Lab (specimens)
- Clinical Site ↔ Patient (in decentralized trials)
- Returns, reconciliation, or destruction chain
This lifecycle requires controlled transportation, real-time visibility, temperature validation, and secure chain-of-custody records at every stage.
For organizations evaluating structured healthcare delivery infrastructure for clinical programs, review clinical trial logistics services.
Clinical Trial Logistics vs. Standard Medical Courier Services
| Clinical Trial Logistics | Standard Medical Courier |
| Protocol-driven | Delivery-driven |
| Chain-of-identity controls | Signature confirmation |
| Audit-ready documentation | Basic proof-of-delivery |
| Temperature deviation response | Temperature transport |
The Core Processes in Clinical Trial Logistics
Clinical trial logistics performance is determined by four operational layers: investigational product distribution, specimen transport, cold chain governance, and documentation control.
Clinical trial logistics functions as the operational engine of the broader clinical trial supply chain, connecting manufacturing, site execution, and laboratory analysis.
Each layer introduces regulatory exposure, timing sensitivity, and measurable risk.
When these processes are standardized and monitored, clinical trial logistics supports protocol adherence and site activation. When they lack structure, small execution gaps can cascade into dosing delays or data loss.
In practice, sponsors often discover documentation gaps only during monitoring visits or audit preparation, when corrective action becomes more complex and costly.
1. Investigational Product (IP) Distribution
Investigational product distribution is the most compliance-sensitive component of clinical trial logistics management. Unlike commercial pharmaceutical shipping, investigational product (IP/IMP) movement is governed by protocol timing, blinding requirements, and sponsor oversight obligations.

Temperature conditions may include refrigerated (2–8°C), frozen, ultra-low, or controlled room temperature. Some therapies are vibration-sensitive or light-sensitive. Others involve controlled substances that require enhanced custody verification.
Operationally mature IP distribution programs include:
- Packaging verification prior to dispatch, including validation configuration review
- Alignment with clinical site dosing schedules and delivery windows
- Controlled handling procedures for restricted products
- Documented chain-of-custody capture with timestamped signatures
- Predefined escalation pathways for transit delays
Unlike retail or enterprise distribution networks, investigational product cannot simply be rerouted or delayed without consequence. A missed delivery window may shift dosing timelines, trigger protocol deviations, or require sponsor review.
For Logistics & Transportation leaders, the takeaway is straightforward: IP distribution is not a routing problem. It is a protocol execution function.
2. Specimen Collection & Clinical Research Logistics
Specimen transport represents the most time-sensitive component of clinical research logistics. Blood draws, tissue samples, and other biological materials must reach central laboratories within defined stability windows to preserve analytical validity.

Specimen logistics failures do not merely delay processing. They can invalidate patient data entirely.
Operational controls in specimen transport typically include:
- Time-bound pickups coordinated directly with site workflow schedules
- Biohazard packaging verification prior to courier release
- Temperature stabilization using validated materials
- Direct-to-laboratory routing to minimize handling and transfer points
Specimen routing should be designed to reduce touchpoints and eliminate unnecessary depot staging when possible. Temperature stabilization, biohazard labeling, and chain-of-custody verification must be confirmed before release from the clinical site. For a deeper operational overview of packaging standards, labeling requirements, and transport controls, see our detailed guide on transporting blood and specimens in healthcare logistics.
From an ICP perspective, this is where clinical supply chain leadership and lab operations intersect. Delays at this layer directly affect data integrity and regulatory defensibility.
3. Cold Chain Management in Clinical Trial Logistics
Cold chain integrity underpins both investigational product distribution and specimen transport. As advanced biologics, cell therapies, and temperature-sensitive compounds become more prevalent, cold chain governance has become central to clinical study logistics.

Programs typically rely on:
- Validated passive packaging systems for short-duration transit
- Active temperature-controlled containers for extended or high-risk shipments
- Continuous temperature monitoring devices
- Defined acceptable excursion thresholds
- Documented quarantine and investigation procedures
- Secure digital retention of temperature data for audit
Cold chain experts consistently report that most temperature excursions stem from preventable failures, including packaging misconfiguration, transit delays, or improper handling during site transfers. A recent industry analysis on temperature excursion risks in pharmaceutical cold chain logistics reinforces that monitoring and rapid exception response are often more decisive than packaging alone.
The operational implication is critical: cold chain is not a packaging decision. It is a monitoring and response framework.
Cold chain governance should be treated as an operational control framework rather than a packaging decision. Monitoring, validation, and documented exception response are what protect investigational product integrity under real-world transit conditions. For a detailed operational overview, review our guide to cold chain logistics in healthcare environments. Clinical & Healthcare Supply Chain leaders should treat temperature compliance as a measurable performance indicator tied directly to protocol adherence and product loss exposure.
4. Documentation, Chain-of-Custody & Proof-of-Delivery
In clinical trial logistics, documentation is regulatory protection.
Every transfer must be traceable. Every handoff must be verifiable. Every exception must be recorded.
Essential documentation typically includes:
- Timestamped pickup confirmation
- Digital signature capture at delivery
- Condition verification upon receipt
- Chain-of-custody logs with custody transitions
- Temperature reports linked to shipment ID
Under the finalized ICH E6(R3) Good Clinical Practice guideline (2025), sponsors are required to ensure appropriate oversight of trial-related processes, including vendor management and essential record retention.
That oversight obligation extends directly to logistics controls.
Digital proof-of-delivery platforms play a central role in demonstrating compliance. Time-stamped confirmations and condition verification provide defensible evidence during monitoring visits or regulatory inspection.
For enterprise logistics leaders, documentation should not be treated as clerical output. It is an audit asset.
The 5 Biggest Challenges in Clinical Trial Logistics
Most vendor pages list challenges in clinical trial logistics. Few explain how those challenges translate into protocol exposure, financial risk, or regulatory vulnerability. In practice, breakdowns in clinical trial logistics management rarely occur as single events. They compound through coordination gaps, documentation lapses, and delayed exception response.
The following risks consistently shape clinical research logistics performance.
1. Multi-Site Coordination Across Distributed Trial Networks
Modern clinical trials span multiple geographies, time zones, and site capabilities. Activation timelines vary widely depending on regulatory review, staffing, and infrastructure maturity. As trial networks expand, operational variability increases.
Industry research highlights ongoing variability in site activation timelines, staffing capacity, and administrative readiness across distributed trial networks, as detailed in WCG’s 2024 Clinical Research Site Challenges Report.
Operational friction often arises from:
- Variable site operating hours
- Rolling site activation schedules
- Protocol amendments mid-study
- Inconsistent packaging readiness at site level
Without standardized clinical trial logistics protocols, each site adapts independently. That improvisation increases documentation inconsistencies, pickup misalignment, and shipment preparation errors.
For Clinical & Healthcare Supply Chain leaders, multi-site coordination is a systems issue, not a scheduling issue.
2. Temperature Excursions and Product Integrity Risk
Temperature excursions occur when investigational products or biological specimens move outside validated stability ranges. In clinical trial logistics, even minor deviations can trigger quarantine procedures or sponsor review.
Common root causes include:
- Packaging misconfiguration
- Unexpected transit delays
- Improper handling during custody transfer
- Inadequate real-time temperature monitoring
Industry reporting on temperature excursion risks in pharmaceutical cold chain logistics emphasizes that preventable process failures account for a significant portion of excursion events.
Financial and regulatory impacts may include:
- Product quarantine and replacement
- Deviation documentation and CAPA review
- Sponsor investigation
- Potential exclusion of affected data
A reactive model is insufficient. Effective clinical trial logistics management requires predefined temperature thresholds, documented escalation workflows, and centralized monitoring oversight.
Temperature compliance is not a packaging choice. It is a governance function.
3. Regulatory Oversight & GCP Compliance Exposure
Regulatory compliance in clinical trial logistics extends beyond FDA inspection readiness. Sponsors must demonstrate ongoing oversight of vendors and maintain essential trial records.
Under the finalized ICH E6(R3) Good Clinical Practice guideline (2025), sponsors are required to implement risk-based quality management and ensure appropriate vendor oversight and essential record retention.
Logistics-related exposure commonly includes:
- Incomplete chain-of-custody logs
- Missing or unverified temperature documentation
- Inadequate proof-of-delivery records
- Delayed deviation reporting
When documentation gaps emerge during monitoring visits, corrective action becomes retrospective rather than preventive.
For enterprise logistics leaders, compliance risk is cumulative. Documentation discipline must be embedded at every transfer point.
4. Last-Mile Timing Risk in Clinical Study Logistics
The final mile often determines whether a trial remains on schedule. Missed delivery windows, failed pickup attempts, or incorrect routing can disrupt dosing schedules and specimen viability.
Same-day recovery capability becomes critical when:
- Sites close unexpectedly
- Weather or traffic delays extend transit time
- Patients reschedule visits
- Protocol amendments alter shipment timing
Unlike retail distribution, clinical trial logistics does not tolerate flexible delivery windows. Dosing events and laboratory cutoffs are fixed. Without rapid rerouting capacity and escalation procedures, timing risk cascades into protocol deviations.
Logistics & Transportation leaders should evaluate whether their model supports real-time rerouting and direct-to-site responsiveness.
5. Exception Handling Without Structured Playbooks
Exceptions are inevitable in clinical research logistics. What differentiates resilient programs is structured response.
Common logistics exceptions include:
- Damaged or compromised packaging
- Label discrepancies
- Site closure upon arrival
- Patient no-show for specimen collection
A controlled exception response framework typically includes:
- Immediate documentation of the event
- Automated notification to site and sponsor liaison
- Defined authority for quarantine or reattempt decisions
- Logged corrective action with timestamped confirmation
- Sponsor visibility within a defined reporting window
Without a predefined exception playbook, each disruption becomes improvised risk. Improvisation increases variability. Variability increases exposure.
In clinical trial logistics, controlled exception management is a quality system, not a contingency plan.
Key Performance Indicators (KPIs) in Clinical Trial Logistics Management
Many vendors promote “visibility” in clinical trial logistics without defining measurable standards. High-performing clinical trial logistics management relies on quantifiable metrics tied directly to protocol adherence, product integrity, and regulatory defensibility.
In regulated environments, measurement is governance.
Core performance indicators typically include:
- Site-specific on-time delivery rate
- Temperature compliance rate
- Exception resolution time
- First-attempt delivery success rate
- Chain-of-custody completion rate
- Documentation accuracy rate
These KPIs protect activation timelines and patient dosing windows while providing defensible evidence of sponsor oversight. A temperature compliance rate without documented deviation response offers little regulatory protection. An on-time delivery percentage without site-level breakdown lacks operational meaning.
For executive Clinical & Healthcare Supply Chain leaders, KPI dashboards should connect logistics performance directly to business exposure. If on-time delivery declines, what is the downstream impact on site activation and dosing continuity? If excursion frequency rises, what is the financial exposure tied to product replacement, deviation reporting, and audit scrutiny?
In clinical trial logistics, metrics are not operational vanity indicators. They are compliance infrastructure.
Clinical Trial Logistics Best Practices for Sponsors and CROs
Awareness of risk does not prevent exposure. Structured controls do. Clinical trial logistics programs require defined governance before first patient enrollment.
Standardize Handoff Protocols Across Clinical Sites
Every shipment should follow a documented, repeatable release protocol. Site-level variability is one of the largest sources of documentation gaps.
Effective handoff checklists typically include:
- Confirmation of labeling accuracy and protocol alignment
- Verification of validated packaging configuration
- Activation and confirmation of temperature monitoring devices
- Condition verification prior to courier release
Consistency across distributed trial networks reduces preventable deviations.
Define RACI Across Clinical Trial Stakeholders
Clear accountability prevents escalation delays.
Programs should formally define:
- Who escalates transit delays
- Who documents and reports deviations
- Who authorizes re-shipments or quarantine
- Who communicates directly with sponsor and CRO
Ambiguity in responsibility introduces response lag. In time-sensitive clinical study logistics, delayed response often carries regulatory consequence.
Build an Exception Playbook Before First Patient In
Exception management should be predefined, not improvised.
Examples of structured thresholds include:
- Transit delay exceeding 30 minutes triggers automated notification to site and sponsor liaison.
- Temperature deviation beyond validated threshold initiates quarantine and sponsor review within a defined reporting window.
- Failed delivery attempt triggers same-day reattempt or approved alternate routing protocol.
Predefined escalation controls convert disruption into documented quality management rather than operational volatility.
Centralize Visibility and Audit-Ready Documentation
Real-time tracking platforms and centralized digital documentation allow sponsors and CROs to maintain oversight across all active sites. Audit data should be searchable, exportable, and retained in alignment with protocol requirements and regulatory standards.
Digital proof-of-delivery systems strengthen this layer of oversight. Clinical trial logistics management becomes resilient when visibility is centralized and exception response is measurable.
Evaluating a Clinical Trial Logistics Partner
Selecting a clinical trial logistics provider requires more than confirming courier availability. In regulated healthcare environments, process discipline and documentation rigor are as important as transit speed.
Evaluation criteria should include:
- Demonstrated experience in healthcare and protocol-driven delivery environments
- Validated temperature management capabilities
- Real-time shipment tracking and reporting tools
- Digital chain-of-custody and proof-of-delivery systems
- Same-day and STAT recovery support
- Trained, vetted drivers with healthcare handling experience
- National U.S. coverage across major clinical markets
Organizations evaluating structured healthcare delivery infrastructure can review healthcare logistics services.
Programs involving laboratory coordination should also assess lab logistics support capabilities.
Clinical trial logistics partners should demonstrate documented process controls, escalation procedures, and measurable KPI tracking — not simply transportation capacity.
How Dropoff Supports Clinical Trial Logistics Programs
Dropoff’s clinical trial logistics model is designed around operational reliability and healthcare compliance rather than generalized courier services.
Programs support:
- Same-day and scheduled investigational product delivery
- Dedicated route programs for recurring specimen pickups
- Digital chain-of-custody documentation with verification
- Real-time tracking and delivery confirmation
- Trained, vetted couriers familiar with healthcare protocols
- 24/7 operational support
- National footprint across major U.S. clinical markets
The emphasis remains on protecting timing, documentation integrity, and regulatory compliance at every transfer point.
When to Strengthen Your Clinical Trial Logistics Model
Clinical trial logistics rarely fails through a single event. It erodes through repeated minor inefficiencies.
Organizations should reassess their logistics framework when:
- Site count increases rapidly across geographies
- Investigational products become more temperature sensitive
- Exception frequency trends upward
- Audit findings cite documentation gaps
- Activation timelines extend without clear root cause
Growth in trial complexity demands parallel growth in logistics governance.
Conclusion
Clinical trial logistics is not an ancillary service. It is a compliance-critical, patient-impacting operational discipline that governs how investigational products, specimens, and essential materials move throughout a study lifecycle.
Strong clinical trial logistics protects:
- Protocol compliance
- Patient safety
- Data integrity
- Activation timelines
As trial complexity increases and regulatory expectations tighten, clinical trial logistics management must be structured, measurable, and audit-ready.
Organizations seeking to evaluate or strengthen their model can request a logistics assessment.
Clinical trial logistics succeeds when every movement is controlled, documented, and predictable. That discipline is what keeps studies on schedule and data defensible.
Frequently Asked Questions About Clinical Trial Logistics
Clinical trial logistics is the coordinated management of investigational products (IP/IMP), laboratory kits, biological specimens, and clinical supplies throughout a study lifecycle. It ensures temperature control, chain-of-custody documentation, regulatory compliance, and on-time delivery between manufacturers, depots, clinical sites, laboratories, and patients.
Clinical trial logistics differs from standard medical courier or clinical trial courier services because it operates under protocol-specific and Good Clinical Practice (GCP) requirements. Unlike standard medical courier services, it requires documented chain-of-custody, temperature validation, blinding protection, deviation tracking, and sponsor oversight. The focus extends beyond delivery speed to regulatory compliance, data integrity, and audit readiness.
Cold chain management protects investigational products and biological specimens by maintaining validated temperature ranges during transit. Temperature deviations can trigger quarantine procedures, product replacement, deviation documentation, and potential data exclusion. Continuous monitoring and predefined escalation workflows are essential to preserve protocol compliance and product integrity.
When a shipment exceeds validated temperature limits, the product or specimen is typically quarantined pending sponsor review. The event must be documented, investigated, and assessed for potential impact on protocol adherence or data validity. Corrective and preventive actions may be required before the shipment is released or replaced.
Key metrics in clinical trial logistics management include on-time delivery rate, temperature compliance rate, exception resolution time, chain-of-custody completion rate, and documentation accuracy rate. These indicators measure operational reliability, protect patient dosing windows, and provide defensible evidence of sponsor oversight.
Investigational product distribution refers to the controlled transport of study drugs or therapies from manufacturers or depots to clinical sites. It requires temperature validation, protocol-aligned delivery timing, documented chain-of-custody, and secure handling to maintain blinding and regulatory compliance.
When selecting a clinical trial logistics provider, sponsors should evaluate healthcare experience, validated temperature management capabilities, digital chain-of-custody systems, real-time tracking tools, documented escalation procedures, and geographic coverage aligned with trial site distribution. Process discipline and regulatory documentation are as critical as transit speed.
Evaluate Your Clinical Trial Logistics Model
As clinical trial networks expand and regulatory expectations tighten, logistics governance becomes a measurable performance driver. Sponsors and CROs that treat clinical trial logistics as structured infrastructure—rather than reactive coordination—reduce deviation exposure and protect activation timelines.
If your organization is assessing temperature controls, documentation rigor, or multi-site coordination performance, request a clinical logistics assessment to evaluate your current model.